September 26, 2022
Tying Together Iron and Carbon Cycling in the Arctic
Iron cycling is key to modeling greenhouse gas emissions from permafrost soils.
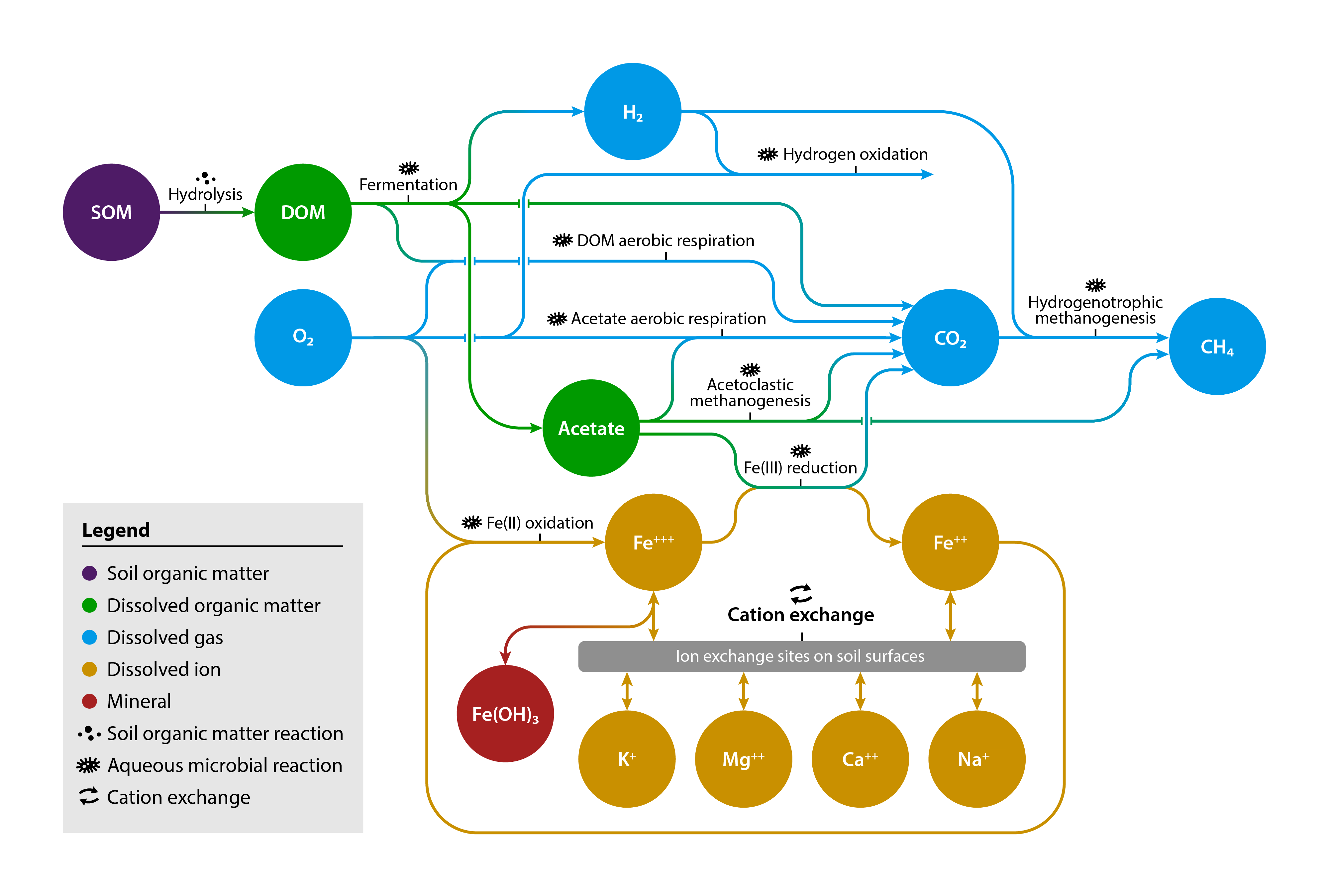
Diagram of the chemical reaction network used to simulate carbon-iron interactions.
[Reprinted with permission from Sulman, B. N., et al. "Simulated Hydrological Dynamics and Coupled Iron Redox Cycling Impact Methane Production in an Arctic Soil." Journal of Geophysical Research: Biogeosciences 127 (10), e2021JG006662 (2022). DOI:10.1029/2021JG006662. © 2023 American Geophysical Union.]
The Science
Emissions of methane, a powerful greenhouse gas, could increase as frozen soils in cold regions thaw. This study used a new computer model to simulate how iron, oxygen, and carbon interact to drive carbon dioxide and methane emissions in waterlogged permafrost soils. Iron-reducing microorganisms used iron to fuel carbon dioxide production, but the effect of iron cycling on methane production depended on availability of easily decomposable carbon. When iron reducers competed with methane producers for a small amount of available carbon, methane production declined. However, when easily decomposed carbon was abundant, iron reduction enhanced methane production by decreasing soil acidity.
The Impact
As frozen soils thaw, carbon within them can be converted to carbon dioxide or methane gas. Because methane has a stronger climate warming effect than carbon dioxide, the relative amounts of the two gases that are produced from decomposition are important to predict the impact of permafrost thaw on climatic warming. Iron is thought to suppress production of methane, but results show that iron could enhance methane production in some soils. This study builds groundwork for improving predictions of Arctic feedbacks to climate change by including iron effects on greenhouse gas production from thawing permafrost.
Summary
Methane production is sensitive to soil acidity. Many Arctic soils are rich in iron, which some soil microorganisms can use instead of oxygen for respiration through iron reduction. This produces carbon dioxide while decreasing soil acidity. Computer models that currently predict greenhouse gas emissions from thawing Arctic soils do not include iron or acidity changes. This study used a chemical reaction network model to simulate interactions of iron reduction, methane production, and organic matter decomposition in permafrost soils. The model was compared to measurements of carbon dioxide and methane production as well as soil acidity from a series of laboratory incubation experiments. The model then simulated cycles of waterlogged and aerated conditions to test how iron affected production of greenhouse gases over multiple cycles. Iron reduction occurred during waterlogged periods, producing carbon dioxide and reducing soil acidity, while iron was recycled during aerated periods. Because methane-producing microorganisms prefer less acidic soil conditions, iron reduction enhanced methane production when there was enough available organic matter to support both processes. When easily decomposed organic matter was more limited, iron reducers competed with methane producers, leading to lower methane production.
Principal Investigator
Benjamin Sulman
Oak Ridge National Laboratory
sulmanbn@ornl.gov
Program Manager
Daniel Stover
U.S. Department of Energy, Biological and Environmental Research (SC-33)
Environmental System Science
daniel.stover@science.doe.gov
Funding
This research was supported by the Biological and Environmental Research (BER) Program in the U.S. Department of Energy’s (DOE) Office of Science as part of the Next-Generation Ecosystem Experiments-Arctic (NGEE-Arctic) project. This research used resources from the Compute and Data Environment for Science (CADES) at Oak Ridge National Laboratory, which is supported under Contract No. DE-AC05-00OR22725. Coauthors also received support from BER’s Coastal Observations, Mechanisms, and Predictions Across Systems and Scales—Field, Measurements, and Experiments (COMPASS-FME) project.
Related Links
References
Sulman, B. N., et al. "Simulated Hydrological Dynamics and Coupled Iron Redox Cycling Impact Methane Production in an Arctic Soil." Journal of Geophysical Research: Biogeosciences 127 (10), e2021JG006662 (2022). https://doi.org/10.1029/2021JG006662.

